Min protein spatial control
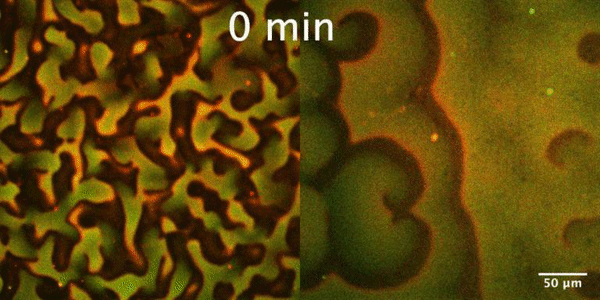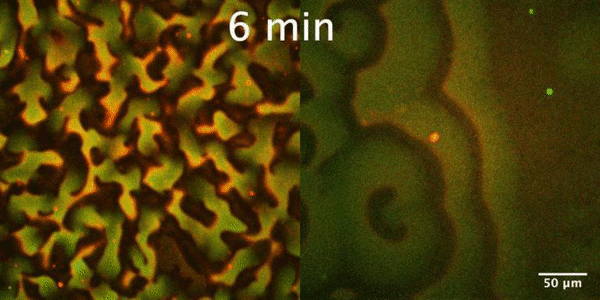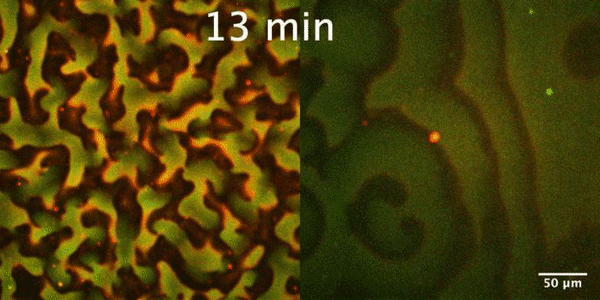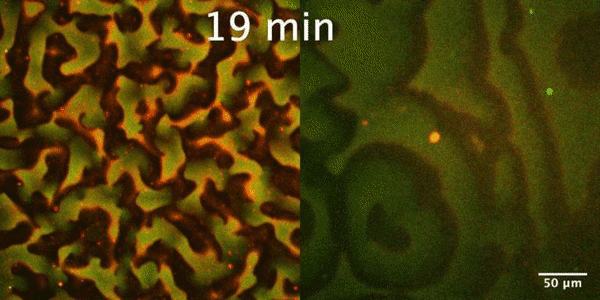
The Min protein system is a molecular reaction-diffusion system that plays an important role in the proper positioning of Z-ring, the membrane-bound contractile polymer that initiates and directs the cell division process in E. coli. Constant pole-to-pole oscillations of Min proteins within E coli cells, which inhibit the formation of the Z-ring, result in the generation of a spatiotemporal concentration gradient with the minimum in the mid cell, that subsequently allows the formation of Z-ring and thus proper cell division.
In our group, we study the Min protein system in a number of in vitro arrangements. We reconstitute Min system using microfluidics in fully confined three-dimensional chambers, rod-shaped water-in-oil droplets and liposomes to understand geometrical selection rules governing Min oscillations in cell-size spaces. Min system behavior is driven by reaction-diffusion mechanism. Changing RD parameters we want to learn how this system is controlled and understand the essence of substantial differences between Min system in vitro and in vivo. Our interest is also focused on understanding mechanism of emergence (symmetry breaking) and evolution of Min patterns. Finally we want to study an interplay between Min system and FtsZ.

People working on this project

Sabrina Meindlhumer
Greg Pawlik
Yaron Caspi

Cees Dekker
- F0.210
- +31-(0)15-27 86094
- C.Dekker@[REMOVE THIS]tudelft.nl
- Principal Investigator
- View CV

